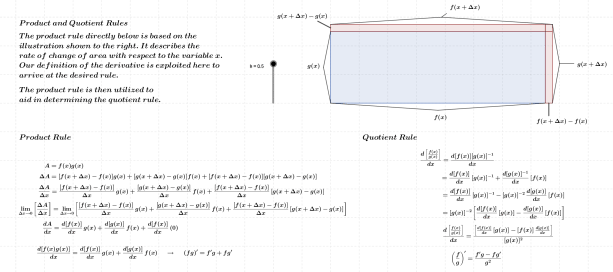
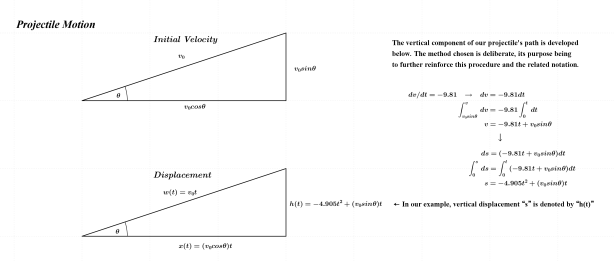
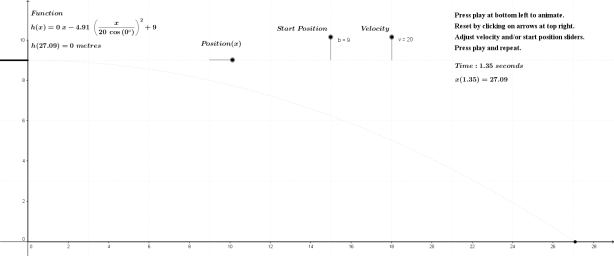
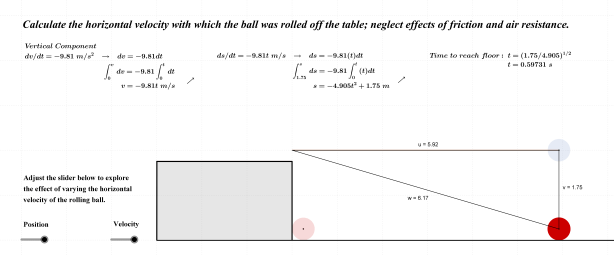
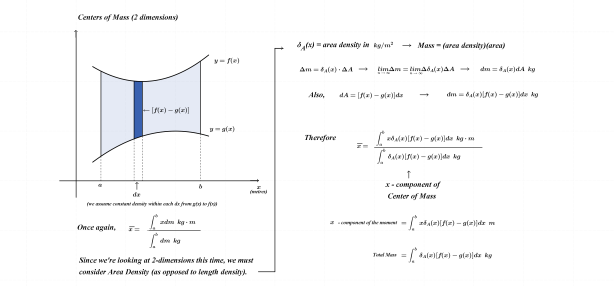
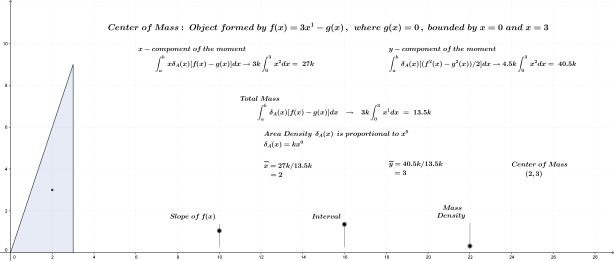
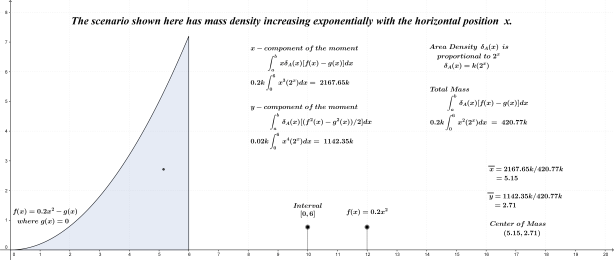
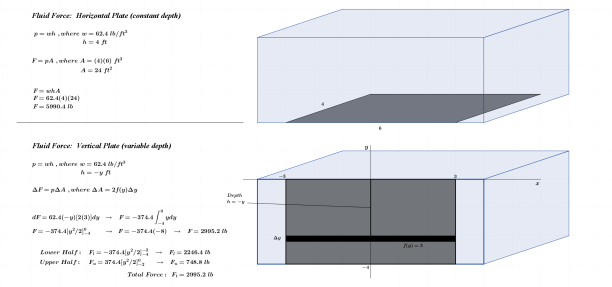
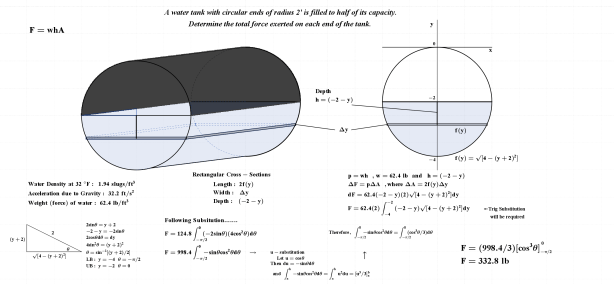
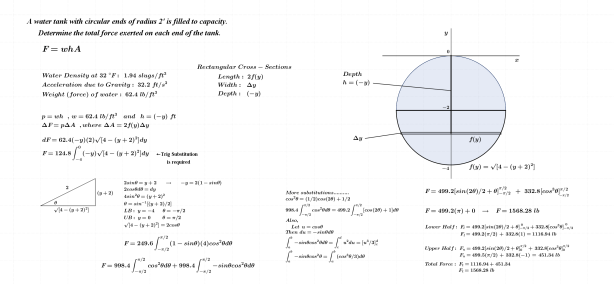
As noted above, the subject of this entry is first order differential equations. Before getting directly to that, however, a few examples of more basic equations representing rates of change will be reflected on.
Too often, students are given rules and algorithms to follow in “solving” problems. While becoming fluent with these procedures is not unimportant, students relying on these strategies alone will eventually find themselves swimming in a pool of confusion when faced with unfamiliar looking problems; if problems not recognizable, they become impossible to resolve. In an effort to minimize such predicaments, an intuitive perspective can often be helpful. Directly below are two examples of solutions to basic differential equations; the first resulting in a quadratic function, the second being exponential. The first two frames below show an “intuitive” approach to solving these problems while the third frame shows a more typical procedure.
Quadratic Function

Exponential Function

Procedure using Leibniz Notation

An intuitive feel is supplementary to the procedures shown above and can add understanding to the process. Equally important is developing the ability to create these mathematical models from information conveyed in the written word.
A huge stumbling block for many students is knowing why a specific model works for one scenario and not the next; the deciding factor is sometimes a seemingly small detail that is easily overlooked early in the learning process. The example below draws attention to this and will serve as a bridge between simple separable differential equations and the more complex first order linear differential equations.
Mixing Problems and Solution Concentrations
This portion of our journey will focus on the changing concentration of a solution in a container as the ratio of solute to solvent increases or decreases. One such example is provided below.
A tank initially contains 50 gallons of brine in which 5 lb of salt is dissolved. Brine containing 1 lb of salt per gallon flows into the tank at a constant rate of 4 gal/min. The concentration of the brine in the tank is kept uniform throughout the tank by stirring. While the brine is entering the tank, brine from the tank is also being drawn off at a rate of 2 gal/min. Find the amount of salt in the tank after 25 minutes.
(Fraleigh, p.903)
In this scenario, the concentration of brine being added to the tank differs from that originally present with the new mixture being drained off simultaneously; this is the “small detail” making the problem here a more complex one than the other examples cited earlier. The two frames immediately below illustrate simplified versions of this and will serve as an introduction to mixing problems; both allow for a straight forward model involving separable differential equations.
In the scenario directly below, the concentration of solute initially contained in the tank has been altered to match that which is being added (1 lb/gal). That being the case, the concentration of solution being drained away will be a constant 1 lb/gal as well; the resulting function will therefore be linear.
Separable Differential Equation (Equal Concentration)

This second example sees the concentration initially contained in the tank revert to 0.1 lb/gal with inflow of 1 lb/gal. This time, however, no solution will be drawn from the container, once again resulting in a linear function.
Separable Differential Equation (Outflow Zero)

First Order Differential Equations
It is now time to tackle the problem posed by Fraleigh. To reiterate, the concentration of solution flowing into the container differs from that initially present AND this new mixture is being drawn off simultaneously, thereby requiring a more complex model. Since the concentration of solution being extracted is constantly changing, that quantity will be expressed as a ratio of salt (in lbs) to number of gallons of brine in the container at any time “t”. Since the quantity of salt in the tank is changing continuously, a variable will be assigned to represent that quantity; we’ll use “y” to denote pounds of salt.
Inflow: 4 gal/min at 1 lb/gal → 4 lb/min Outflow: 2 gal/min at y lb/(50+2t) gal → [2/(50+2t)]y lb/min
From this, we can easily represent the rate of change of salt (in lbs) with respect to time as follows:
dy/dt=4-[2/(50+2t)]y (lb/min)
This expression is referred to as “Item 2” in the frame directly below. The “item” preceding that is a reminder of the FTC that is called upon in solving for what is referred to as an “Integrating Factor”. Its purpose is also discovered below and is denoted by “I”.
Integrating Factor

Purpose of the Integrating Factor (clarification)
Given Iy’+Ipy=qI
If (Iy)’=Iy’+Ipy , then (Iy)’=qI which leads to Iy=∫qIdt
The integrating factor ultimately allows us to express “y” as a function of “t” (which is our objective).
While first order differential equations are not required in cases where the concentration of inflow equals that already in the container, initial conditions for that scenario are nevertheless included below. The solution to Fraleigh’s problem is shown as well (bottom right) for comparison purposes.
Solution to Fraleigh’s Problem

To reiterate: If the concentration of solution flowing into the container is equal to that already present OR outflow is zero, the function describing quantity of solute will be linear in nature. The following three frames serves as examples of this important detail.
Linear Function: Constant Concentration (1 lb/gal)

Linear Function: Constant Concentration (2 lb/gal)

Linear Function: Zero Outflow

The frame that follows is a variation of the problem posed by Fraleigh, this time with an inflow of 5 gal/min. A graph of the resulting non-linear function is also included.
Non-Linear Function

For further comparison and analysis, I’ve included two additional scenarios directly below, each with an interactive link.
Inflow Exceeds Outflow

Click on the link provided here to explore scenarios where inflow exceeds outflow.
Outflow Exceeds Inflow

Click on the link provided here to explore scenarios where outflow exceeds inflow.
The following image illustrates the function shown directly above on a different scale. The lack of symmetry here creates additional opportunities for discussion with respect to the rate at which the quantity of solute is changing over time. For example, at t=11.16 minutes it can be stated that the quantity of solute flowing in equals that which is flowing out. What is happening before and after that point in time and why? Is this a reasonable conclusion and does the graph of our function reflect that?

Nearly the end………….
I had initially planned on ending this entry with the previous frame; my students, however, wanted one more day to explore mixing problems. I’ve decided to summarize that final day’s sequence here.
As always, this mixing problem began as follows:
y’=q-py , where y’ = rate of change of solute (lb/min) q = inflow of solute (lb/min) py = outflow of solute (lb/min)
We then decided initial conditions, concentration of solute being added as well as rates of inflow and outflow. The first set of values chosen produced what we thought was a reasonable solution until that function’s stationary point was determined; it occurred at (31.53,-1.96). These values caused some concern so our work was double checked. Since the math seemed to be fine, we decided to graph our function and interpret that. Once everything was taken into consideration, this result was perfectly logical. The tank initially contained 150 lbs of solute (3 lbs/gal); we were adding only 0.2 lb/gal at a rate of 6 gal/min and draining away the diluted mixture at a rate of 8 gal/min. Upon reflection, our result seemed perfectly reasonable. This scenario and resulting function are shown immediately below.
Rapid Dilution

Following our satisfactory explanation of the dilution scenario above, the discussion focused on what would need to change so that the function would produce a maximum value greater that 150 lb (the initial content). It was decided that increasing the concentration of solute flowing in would produce the desired result. A concentration of 4 lb/gal was agreed upon and the function reworked. Upon graphing this new function, the location of its maximum was revealed. The question immediately asked was ‘Why is the maximum located at t=0?’. The derivative was determined and set equal to zero, quickly satisfying everyone. The frame immediately below illustrates this scenario.
Local Maximum at t=0

To further solidify our understanding of this concept, it was decided that the vertex of our graph should move to the right if the concentration of solute flowing in was further increased. This final iteration of our mixing problem is shown in the following frame.
Local Maximum at t>0

Click on the link provided here to further explore mixing problems.
The end.
Thanks for reading.
References
Kline, Morris. (1998). Calculus: An Intuitive and Physical Approach. Mineola, NY: Dover Publications.
Fraleigh, J. B. (1985). Calculus with Analytic Geometry. Reading, Mass.: Addison-Wesley.